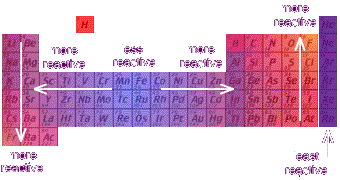
Reactivity and Electronegativity
Electronegativity (Pauline scale)
Measure of an atom's ability to attract electrons in a chemical compound
Reactivity
Group 13Metals:
Reactivity increases as you go down the group. ** The farther to the left and down the periodic chart you go, the easier it is for electrons to be given or taken away, = higher reactivity. Non-Metals: Reactivity decreases as you go down the group. ** The farther right and up you go on the periodic table, the higher the electronegativity, = harder exchange of electron. |
Period 7Metals:Reactivity decreases as you go from left to right across a period.
Non-Metals: Reactivity increases as you go from the left to the right across a period. |