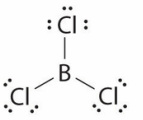
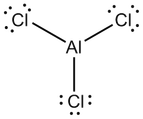
Valence Electrons and Lewis structures
Valence Electrons
Outer shell electrons involved in bonding
Group 13B - 3
Ac - 3 Al - 3 Ga - 3 In - 3 Tl - 3 **Trend: All of the elements have 3 valence electrons |
Period 7Fr - 1
Ra - 2 (f block has 1 or 2 valence electrons) Ac - 3 Rf - 4 Db - 5 Sg - 6 Bh - 7 Hs - 8 Mt - 8 |
Lewis Structures
Group 13
Because each of the elements have the same number of valence electrons, their Lewis Structures have the same shape when bonded with the same element.
|
Period 7
Most of the elements in this period have either one or two valence electrons. Despite the transition metals having an uncertain number of valence electrons, they will most likely give away [rather than take] or share electrons.
|