Ionization Energy and Electron Configurations
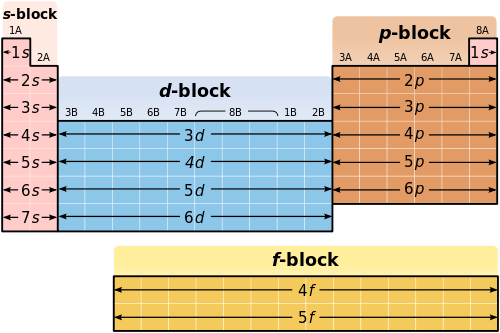
Electron Configurations
Group 13The electron configurations of the elements in Group 13 all end with one electron in p-shaped orbitals.
Examples: Boron=1s^2 2s^2 2p Gallium=1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^10 4p |
Period 7The electron configurations of the elements in Period 7 all end in either the 7s, 6d, or 5f orbitals.
Examples: Francium=1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^10 4p^2 5s^2 4d^10 5p^6 6s^2 5d^10 6p^6 7s Seaborgium=1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^10 4p^25s^2 4d^10 5p^6 6s^2 4f^14 5d^10 6p^6 7s^2 5f^14 6d^3 Americium=1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^10 4p^25s^2 4d^10 5p^6 6s^2 4f^14 5d^10 6p^6 7s^2 5f^7 |
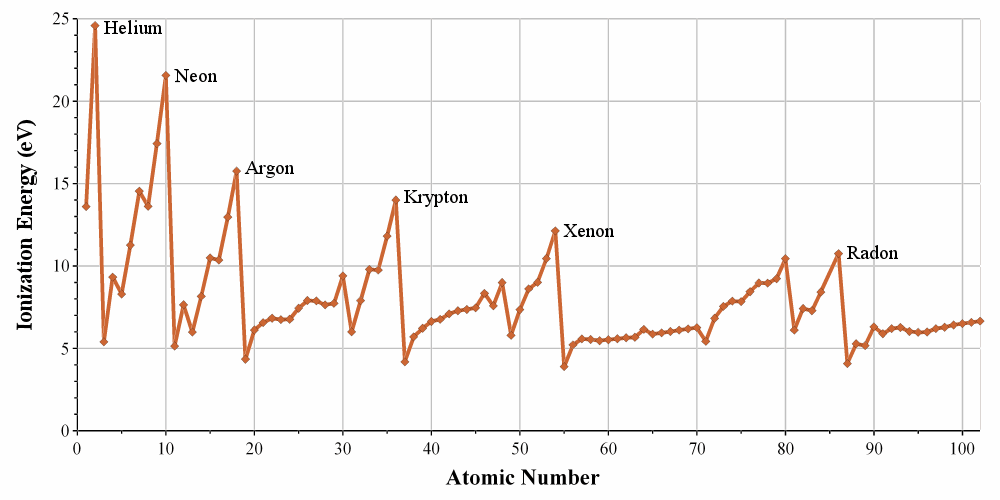
Ionization Energy (kilojoules per mole ----> kJ/mol)
energy needed to remove one atom
**in general, increases across a period and decreases down a group
**in general, increases across a period and decreases down a group